What is a Battery? A Simplified Expert Explanation
Author:admin Date: 2025-03-10 07:49 Views:178
Introduction
Many electronics are in our homes or offices, and they need the right power source to function. One of those power sources is a battery. Expect to encounter different battery types because these electronic devices have different power needs and battery compartments.
So, what is a battery?
A battery is a device containing active chemical materials that can be converted into electrical energy through an electrochemical reaction called oxidation-reduction or redox. During the electrochemical process, electrons are transferred from one side of the material to another through an electric circuit.

Key Components of a Battery
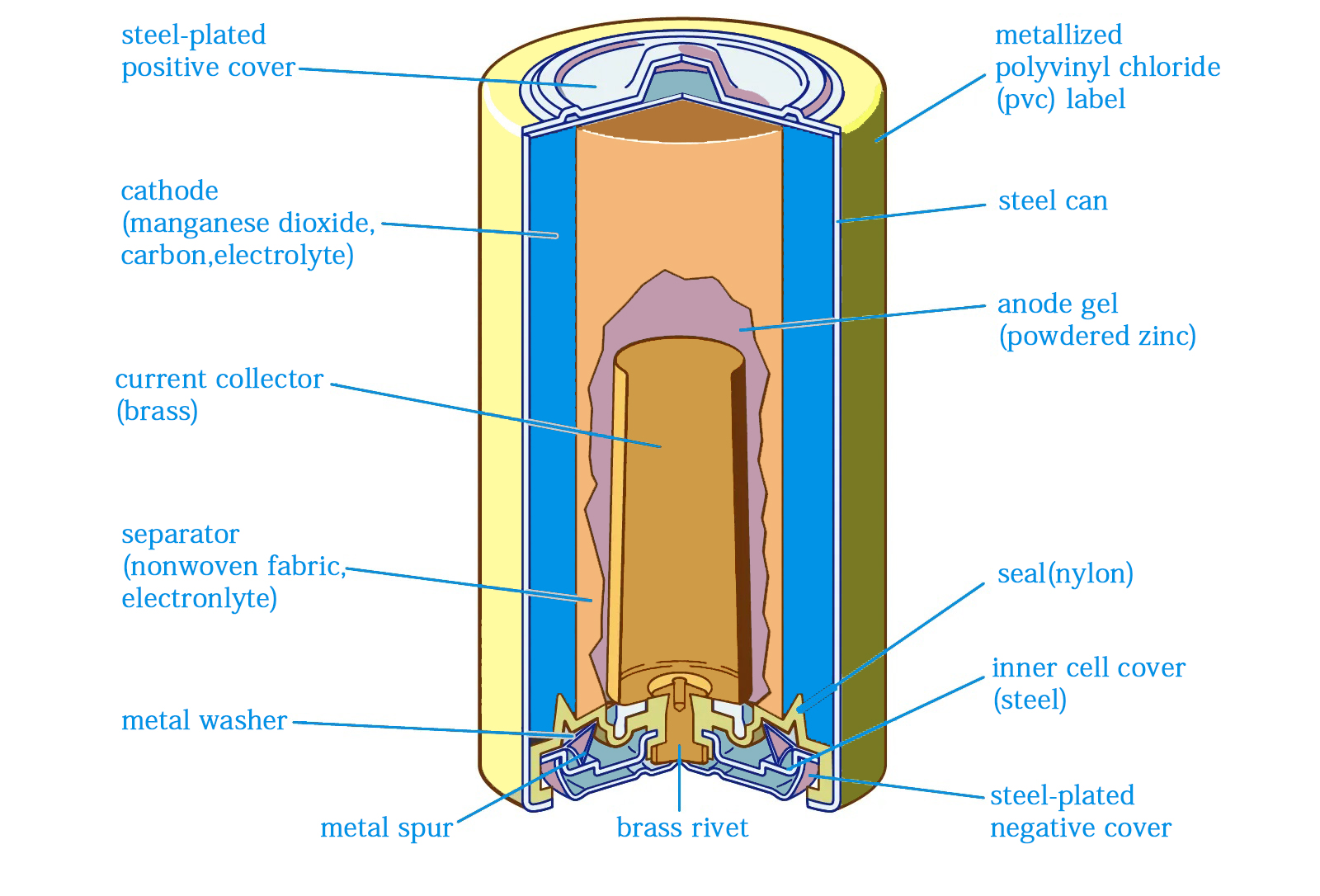
Expect to come across different battery components depending on the battery type. However, some are standard; you should expect to find them in a battery. Here are the key components of a battery:
Cathode
This is the positive electrode of the battery, also called the oxidizing electrode. Its job is to acquire the electrons during the electrochemical process in the battery. Since this is where the reduction process occurs, it will determine the battery’s technology or chemistry. That includes lithium-ion, NiMH, or alkaline.
Anode
This is the battery’s negative electrode. It is where oxidation occurs, meaning it releases electrons during the discharge process. Expect the anode to be made of an electrically conductive material, such as metal.
Electrolyte
Electrolytes are a medium that allows ions to move between the anode and cathode parts of the battery. They come in different states. For ionic conduction, you can have liquid electrolytes such as acids, alkalis, and more, but you can still get solid electrolytes in other types of batteries, including AA, AAA, and more.
Separator
This is a porous material that prevents the anode and cathode sides from mixing. Since it is porous, it allows only ions to move from one side to another, not the materials. If the anode and cathode were to mix, it could lead to the battery exploding or catching on fire.
Terminals
Still, on the subject of batteries and their components, we have the terminals. These are the metal contacts that connect the battery to a circuit. Most batteries have only two terminals located on either side of the battery. It all comes down to the battery design.
How a Battery Works

When you connect a battery to an external circuit, several chemical reactions occur for it to supply electrical energy to the circuit.
The chemical process is called redox, where the electrons are generated and flow from the anode. They are then received and reduced by the cathode. The circuit is complete with the movement of anions and cations in the battery’s electrolyte.
Types of Batteries in the Market
Batteries are classified based on their chemistry. The main classifications are primary, secondary, reserve, and fuel cells.
Primary cell/battery

A primary cell or battery cannot be recharged after it no longer powers the electric circuit. You must discard it after use and replace it with a new battery.
They are mostly called dry cells because they have a solid electrolyte. Manufacturers usually put the electrolyte in an absorbent material that acts as a separator.
Secondary cell/battery

This is the opposite of the primary cell, as it allows for recharging if the capacity decreases. Once you recharge, it goes back to its pre-discharge condition. This is done by passing the current in the opposite direction that you did during discharge.
There are also two subcategories under secondary batteries. The first one is batteries designed to deliver energy on demand. They are always connected to primary power sources and kick in during emergencies.
The other subcategory is batteries that can be used as the main power source and then recharged when their capacity falls below a certain threshold.
Reserve cells/batteries

In this case, the battery’s electrolyte is isolated compared to the primary and secondary cells. The electrolyte remains inactive until a certain melting point allows for ionic conduction. This is what activates the battery and allows it to supply electrical energy to the circuit.
Reserve cells are in heat-activated, gas-activated, electrolyte-activated, and water-activated batteries. They are also found in sensitive detonative devices such as missiles and other weapon systems.
Fuel cell

These batteries are designed to generate electricity only if all the components are fed into them. Unlike the other batteries above, which already have the components, in this case, you have to keep feeding the active materials to the electrodes to keep the battery active. The moment the feeding process stops, the production of electrical energy stops.
A good application for fuel cells is the cryogenic fuels used in space vehicles. Although their applications are limited, their interest has been renewed over the last few years.
Applications of Batteries
Many devices depend on the use of batteries to keep them powered. This means there are so many applications where you can use batteries. We will look at them below to get a good understanding.
As expected, we use batteries in our homes and offices every day. This includes powering mobile phones, remote controls, game consoles, laptops, and more. As long as it needs a battery to work, there is one for it.
These batteries can also be used in health instruments. For example, you need batteries in hearing aids, insulin pumps, and devices with valve assistance that require batteries to function.
Still, in the medical sector, batteries can be used for an ECG heart monitor to show the patient’s vitals. As you keep exploring, expect to come across more applications for batteries in the medical space.
The logistics and construction sectors also use batteries, including heavy-duty batteries. An example is using them in forklifts since they would be used in confined spaces, and using gasoline can lead to carbon monoxide fumes.
Batteries can be used more in firefighting and emergency response. This is where large batteries are used to power various devices that the emergency response teams need to handle emergencies.
We must not forget how batteries can be used in vehicles. Batteries are vital in starting and running accessories. Still, we have electric vehicles that are fully powered by batteries.
Battery Lifespan and Maintenance
Batteries have different lifespans depending on battery chemistry and the load connected to such a battery. Manufacturers often give the estimated lifespan of the battery depending on how they recommend you to use the battery. Expect an average lifespan of 3 to 5 years for most batteries.
You can do a few things to get more life out of your battery. That is why we discuss a few maintenance tips below to keep in mind.
- Regular cleaning of the battery terminals can help prevent corrosion. This ensures proper electrical connections each time the battery is used.
- Follow the manufacturer’s recommendations for discharging and charging. This will ensure that battery capacity remains the same over the years.
- Temperature control is key to determining how well the battery works. Keep the battery in a cool, dry place, away from extreme temperatures.
- Regular inspection is vital to determining when to replace the battery. If you notice the battery bulging or leaking, it could be time to do so.
- If the battery needs a top-up for its acid, make sure it is done on time. It is best to devise a maintenance schedule to ensure you always handle small details that can affect the battery’s performance.
How to Transport Batteries
For certain applications, transporting batteries can be necessary. Because of this, you have to do it right.
The most recommended safety measure is to protect the terminals. Since you would be transporting over long distances, the last thing you want is to end up with short circuits. So, cover the terminals using terminal covers or electrical tape.
Still, consider using sturdy outer packaging to prevent the battery from crushing. You could add plenty of filler where necessary to keep the batteries from constantly shifting during handling.
Clear labeling is also recommended. This helps those handling the batteries know they need extra caution. Sometimes, hazard or fragile warnings may be necessary to encourage people to handle the batteries carefully.
Can Batteries Be Restored?
You can simply recharge a rechargeable battery and continue to use it. However, it is not always possible. Sometimes, the battery wears to a point where it does not hold the capacity as before. It could be that it is old or facing internal damage you do not know about.
If the battery experiences sulfation, it can be revived using pulse charging techniques. Sulfation occurs when sulfate crystals form on the battery’s plates, making it impossible to recharge.
The alternative is to take it to a battery reconditioning center. Let the professionals deal with the process of recycling the battery if they find it still in good condition to keep it running.
Conclusion
A battery can do a lot in our lives today. You may end up having a good time by powering your favorite device and listening to music or using the same to run your vehicle. The applications are quite many. It all depends on your battery needs. Device manufacturers will recommend the battery type you can use for a device. Make sure you use the correct one if you want to end up with the best performance. We still recommend taking good care of your battery if you hope for it to last a long time and give you the best service.
Please send RFQ , we will respond immediately.
Frequently Asked Questions
What is an AGM battery?
This is a valve-regulated lead-acid battery whose electrolyte has been absorbed into a specialized glass fiber mat called an Absorbent Glass Mat. This construction makes the battery leak-proof, maintenance-free, and spill-proof.
What is a lithium battery?
This rechargeable battery type features lithium ions for storage and release of energy. Expect such a battery to have a long lifespan, high energy density, and work with multiple devices.
What voltage is a car battery?
A standard car battery will have 12 volts as the voltage. The voltage will fluctuate depending on the state of charge or when the engine runs.
What is a deep cycle battery?
This is a battery type that can be discharged and recharged repeatedly by providing power over long periods. Expect it to be discharged to much lower states and recharged to be used again. This is unlike vehicle batteries which are designed for offering short bursts of high current.
What is a button battery?
Button batteries are also called cell or coin batteries. Just as the name suggests, they are shaped like a coin with a shiny surface. They are mostly used in small electronic devices such as remote controls, calculators, and watches.
What is a solid-state battery?
For this type of battery, there is the use of solid electrolyte rather than the gel or liquid electrolyte. These batteries are characterized for having high energy density, longer lifespan, and improved safety since there are less risks of fire and leaks.
What is the CCA on a battery?
CCA means cold cranking amps and it is the ability of a battery to deliver enough current to start an engine during the cold temperatures. This figure is mostly noted on the car batteries due to its importance to start a car whenever you need it.


